Hereditary Cancer
Overview of Genetics and Inherited Cancers
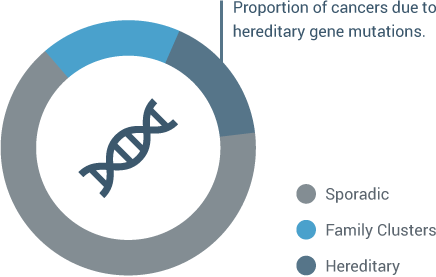
Most cancers occur in patients who do not have a strong family history associated with the inheritance of a single gene mutation. This is often called “sporadic cancer.” In some families we see more cancer than we would expect by chance alone. Determining which of these families have cancer related to an inherited gene mutation is important, as the cancer risks in hereditary cancer families are much higher than the general population. In fact, about 10% of cancers are attributable to a hereditary cancer predisposition. Research has shown that genetic mutations can significantly increase patients’ risks of developing secondary cancers. (Lu et al.) (1)
Understanding these hereditary cancer risks will allow you to recommend more precise medical treatments as well as risk-reducing management options to patients and their family members.
Making hereditary cancer testing standard of care in a practice can help clinicians avoid a misdiagnosis and potentially lead to better outcomes for patients.
“Genetic factors are a key component of precision medicine because they can unlock important information that can help an oncologist determine the best course of individualized treatment,” said ASCO President Clifford A. Hudis, MD, FACP.” An adequate family history is key to identifying those patients whose cancer may be associated with inherited genetic factors.” (2)
Breast and Ovarian Cancers
A woman’s risk of developing breast and/or ovarian cancer is greatly increased if she inherits a deleterious (or pathogenic) mutation in the BRCA1 gene or the BRCA2 gene. Today, the National Cancer Institute reports that BRCA1 and BRCA2 mutations account for approximately:
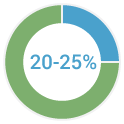
of hereditary breast cancers
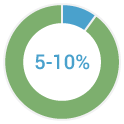
of all breast cancers (3)
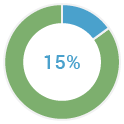
of all ovarian cancers (4)
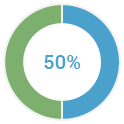
of families with multiple cases of breast cancers
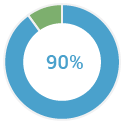
of families with both breast and ovarian cancer
Increased Risks of Breast Cancer
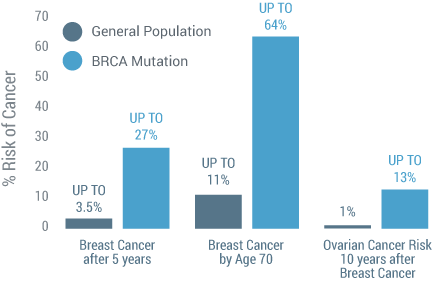
12% of women in general population will develop breast cancer in their lifetime.
Patients with HBOC Syndrome have up to a 27%(5) chance of developing a second breast cancer within 5 years of their initial breast cancer diagnosis, compared to 2%(6) chance for the general population. By age 70, the risk increases to 64%(7).
Patients with a BRCA1 or BRCA2 mutation have an increased risk of up to 13%(8) of developing ovarian cancer within 10 years of their initial breast cancer diagnosis compared to a 0.7%(9) risk for the general population.
BRCA Mutations Increase Risk of a Second Cancer
Colon Cancer and Lynch Syndrome
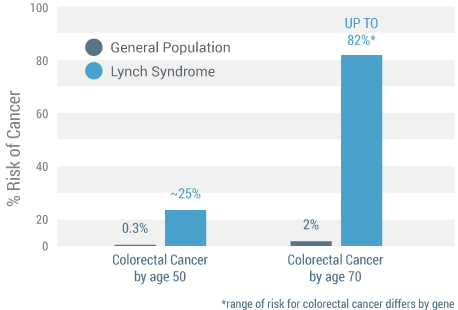
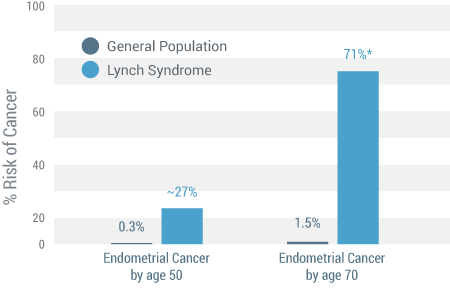
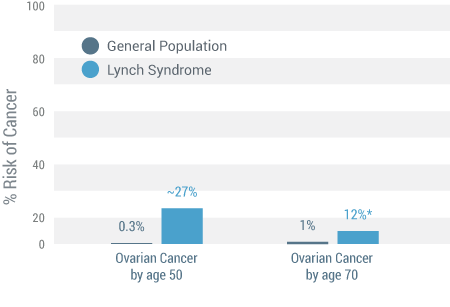
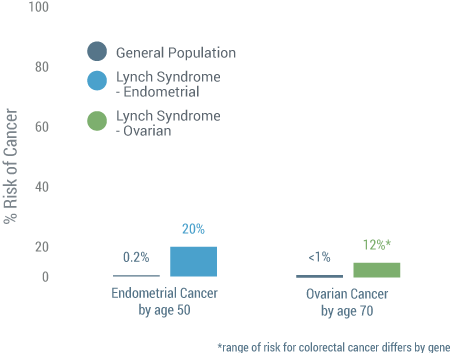
Lynch syndrome, also known as Hereditary Nonpolyposis Colorectal Cancer (HNPCC), is the most common of the hereditary colon cancer syndromes. Lynch syndrome is caused by mutations in the MLH1, MSH2, MSH6, PMS2 or EPCAM genes and is believed to account for 3% to 5% of all colorectal cancers.(10) Patients with Lynch syndrome have greater risks of developing other cancers before the age of 70 years, including:
- Colorectal – 82% versus 2% of general population (11)
- Endometrial – 71% versus 1.5% of general population (12)
- Ovarian – 12% versus less than 1% of general population (12)
Colorectal Cancer (11)
Endometrial Cancer (12)
Ovarian Cancer (12)
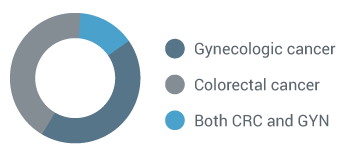
Lynch Syndrome Increases Gynecologic Cancer Risks (13)
Women with Lynch syndrome may present with gynecologic cancer first.
 (12)
(12)
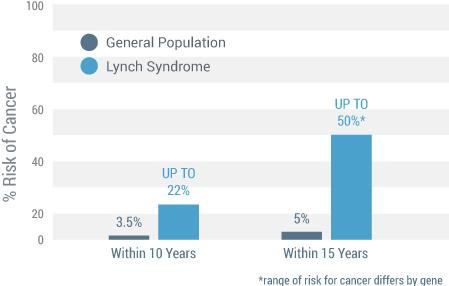
Patients with Lynch syndrome have up to a 30% increased risk for developing a second cancer within 10 years of their initial colorectal cancer diagnosis.
That risk increases to up to 50% within 15 years for a second cancer diagnosis versus the general population’s risk of 5%.
 (12)
(12)
Lynch Syndrome Increases Risk of a Second Cancer
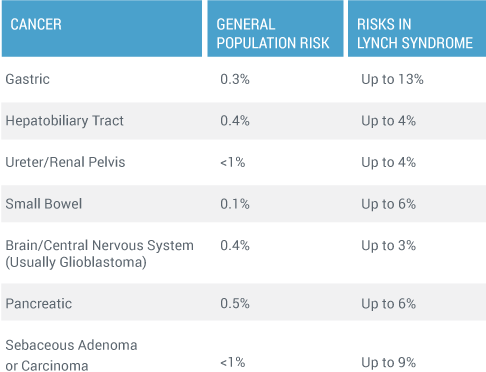
Patients with Lynch syndrome are also at a higher risk of developing gastric, urinary tract, renal/pelvic, biliary tract, small bowel, pancreatic and brain cancers. (14)
Impact on Patients
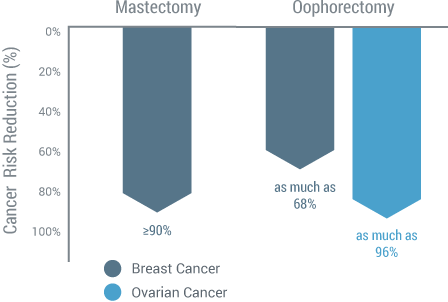
A positive hereditary cancer test result is an opportunity for you and your patients to make informed medical decisions about reducing their cancer risks and planning for the future. Patients with hereditary cancer are candidates for increased screening as well as treatment options including prophylactic surgery and chemoprevention.
(16)
Knowledge Affects Treatment Choice
Research(15) indicates that knowledge of one’s BRCA1 and BRCA2 mutation status before surgery influences a woman’s treatment choice.
Additionally, different treatment options may be recommended to help mitigate the risk of a first or second cancer.
“Patients with hereditary cancer are candidates for increased screening as well as treatment options including prophylactic surgery and chemoprevention.”
Results Encourage More Screenings
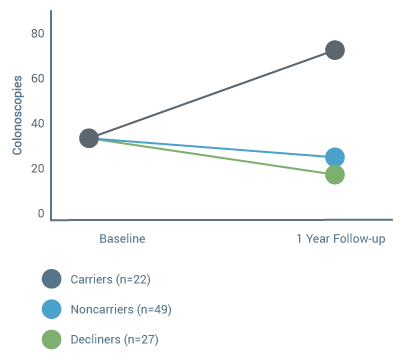
Genetic test results may motivate patients with hereditary gene mutations to undergo more cancer screening tests compared to patients who do not carry a genetic mutation.
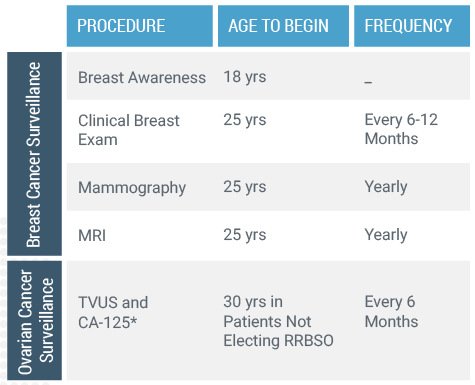
This clinical summary shows data that indicates mutation carriers were significantly more likely to have a colonoscopy than noncarriers and patients who declined testing. Currently, patients with Lynch syndrome are advised to have a colonoscopy every 1 to 2 years starting between the ages of 20 to 25 and annually after age 40.
BRCA Mutation Carriers and Surgical Management (17)
Changes in Colonoscopy Use Following HNPCC Genetic Testing
Management Considerations Change
Consider the differences in management for a 62 year old woman with Stage 2 ER/PR +, HER2 breast cancer. Upon diagnosis, this patient received a lumpectomy, radiation, chemotherapy and endocrine therapy, but a thorough hereditary cancer history was not obtained. After 5 years there was no evidence of disease, so she entered the survivorship program. However, at 7 years she presented with abdominal pain and bloating. A PET showed a mass on the left ovary, she had elevated CA-125, and was diagnosed with Stage 3 ovarian cancer. Additionally, at this point a hereditary cancer risk assessment was completed and it was determined that she met guidelines for further genetic testing. This patient was tested and the results revealed she was BRCA+. Unfortunately, she died of ovarian cancer and this outcome may have been avoided if hereditary cancer was worked up with the same level of importance as recurrence risk on the initial visit. Had this patient’s hereditary cancer status been determined at her initial diagnosis, her provider may have recommended a bilateral mastectomy to reduce her risk of a second breast cancer as well as the possible recommendation of a bilateral salpingo-oophorectomy to reduce her risk of ovarian cancer. Unfortunately, because hereditary cancer status was not part of this patient’s initial diagnosis, the results were fatal.
Patients Tell Their Stories
Hear stories from patients on how their lives changed after receiving a positive hereditary cancer test result:
Copyright © 2015 Myriad Genetic Laboratories all rights reserved