Test Accuracy Importance
Lab Quality Matters
When it comes to hereditary cancer testing, accuracy is EVERYTHING. There are many choices, but accurate results should be the most important factor when selecting a laboratory.
Why It Matters
Labs can make mistakes that negatively affect the accuracy of test results. For example:
- Lab misses DNA changes due to poor test design or not testing for all known mutations.
- Lab incorrectly classifies a DNA change as a change that causes cancer. All patients have DNA changes, but not all changes cause cancer. DNA changes must be assessed carefully and multiple lines of evidence should be used to determine if the changes affect cancer risks.
- Lab lacks a lifetime commitment to update patients and providers with new information that could impact future medical management.
Inaccurate test results can have direct consequences for you and your patients:
- A false negative test result could mean a patient must battle a new cancer in a few years that could have been prevented with appropriate medical management.
- A false positive test could result in unnecessary procedures and wasted healthcare dollars. At worst, a patient could have an unnecessary surgery to remove healthy tissue.
- An inaccurate test result impacts the families of patients. These family members would have inaccurate information about the family’s cancer risks.
The Myriad Difference
Myriad has tested more than 1.5 million patients over the past two decades. A pioneer in molecular diagnostics, Myriad has done more genetic testing THAN any other lab in the world, and has spearheaded important scientific discoveries.
Numerous publications have validated the accuracy of Myriad tests: (1)
- >99.92%: Accuracy of test to find DNA changes
- >99.98%: Accuracy of BRACAnalysis
- 99%: Accuracy of process to assign cancer risks to DNA changes
In the largest validation study ever published for BRCA1 and BRCA2 genes, researchers concluded that optimization of BRCA1 and BRCA2 testing combined with rigorous quality standards at Myriad Genetic Laboratories result in superior analytic sensitivity by either Sanger sequencing or Next Generation Sequencing (NGS).
Additionally, Myriad offers BRACAnalysis CDx, an FDA-approved companion diagnostic, which required a rigorous review by the FDA to ensure the highest lab quality.
It’s a common misconception that Clinical Laboratory Improvement Amendments (CLIA) regulates tests. CLIA regulates laboratories, not the tests that are developed and produced by laboratories. Additionally, CLIA defers regulatory guidelines for clinical laboratories to other professional organizations, such as College of American Pathologists (CAP).


For a test performed in a CLIA accredited lab, if the test is updated, no review for insurance for the continued accuracy of the test will take place before the scheduled inspection, which occurs only once every two years. In contrast, the FDA will review any updates to a test immediately before the update is commercialized. Likewise, validation studies and changes in the test menu are not immediately evaluated by CLIA, while the FDA will block these changes to a commercial test until they have conducted review and approved any changes.
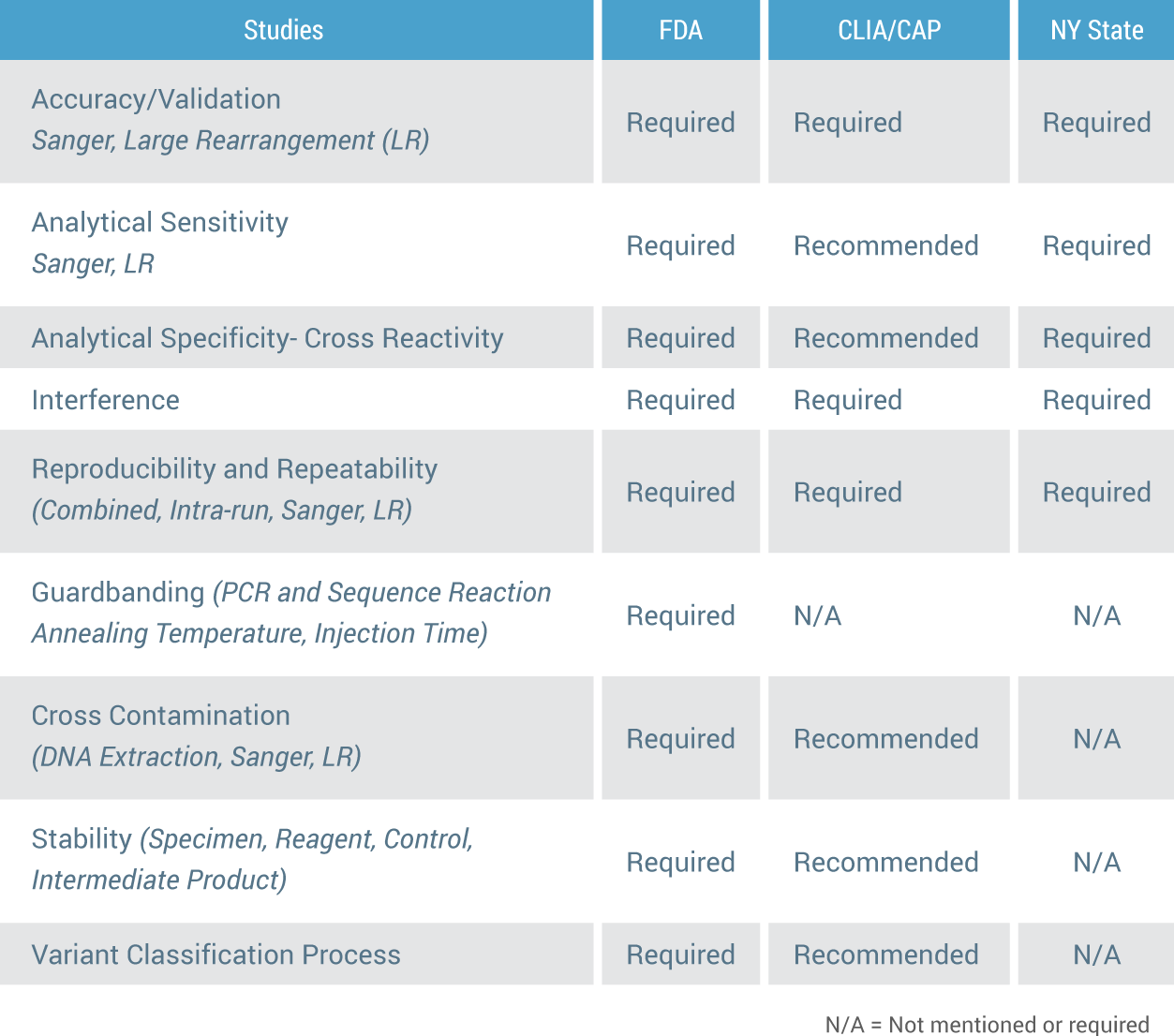
Listed below is a further break-down of the differences in oversight and review between FDA and CLIA. This is a list of the studies required for FDA approval and whether they are required by CLIA/CAP and also by NY State. You can see by this comparison that there is a significant difference between FDA, CLIA/CAP and New York State oversight. Only FDA has standards that are required for all 9 components of a DNA test.
FDA Approved Tests Result in a Higher Standard
We can get a better idea of the difference between CLIA and FDA approval, and why BRACAnalysis CDx was held to the highest standard by enumerating the requirements for each type of clearance. First and foremost, the FDA requires validation of a test’s accuracy and reproducibility; all components of the assay, including equipment, software and processes were validated. On top of this, Myriad’s variant classification process, which is unique in nature compared to other laboratories, was reviewed by the FDA and is part of the approved device.
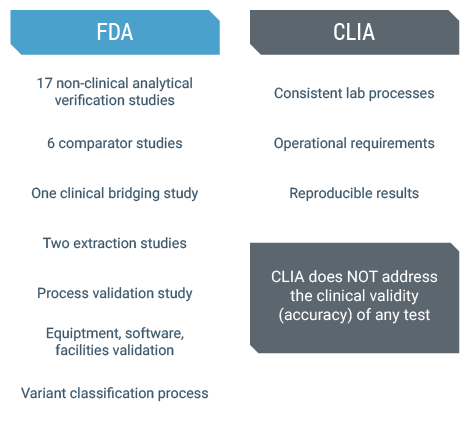
It is important to note that only the FDA review process addresses the clinical validity (or accuracy) of a test whereas other review processes do not.(2)
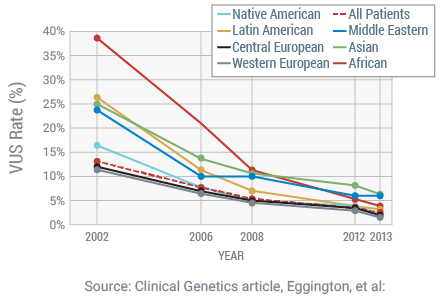
Reducing VUS Results
Myriad has a lifetime commitment to provide patients and providers with the most accurate results and to work diligently to reclassify variants of uncertain significance (VUS) on a weekly basis. A VUS occurs when there is not enough evidence to determine if a genetic change is associated with an increased cancer risk or not. This is a particularly challenging result for patients and providers to receive, because it does not provide a definitive direction on medical management decisions.
Myriad has invested heavily in understanding the significance of new variants. As an example, the BRCA1 and BRCA2 VUS rates are industry-low at 2.1%.
Myriad’s myVision™ Variant Classification Program uses numerous lines of evidence to classify variants. In addition, a diverse team of more than 30 scientific experts review variants as part of the Myriad Variant Classification Team. By tapping into the expertise of these individuals from all applicable disciplines, Myriad’s myVision represents unrivaled experience and expertise in variant classification.
In addition to our team of scientists, Myriad is the only lab that uses multiple lines of validated evidence to classify and reclassify variants. Myriad’s unique clinical history-weighting algorithm, Pheno®, is based on our company’s immense testing volume over the years. Pheno has been validated to be >99% accurate(4) when reclassifying variants. Together with Myriad’s classification method Mutation Co-occurrence, these tools account for more than 60% of reclassifications done at Myriad.

Myriad’s commitment to provide the most accurate testing doesn’t stop when a patient’s results are in hand. We understand that a genetic test result influences the clinical path of a patient. The myVision™ program is part of Myriad’s commitment to patients and their families that lasts a lifetime.
- Myriad offers targeted genetic testing at no cost to selected family members in order to gather more information about a VUS result to assist in the reclassification process.
- When an uncertain variant result is reclassified, an amended report is made available to the provider.
- Myriad continues to evaluate additional information as it emerges in order to classify and reclassify variants quickly and accurately. Leading cancer experts from Memorial Sloan Kettering Cancer Center, Mayo Clinic, Dana Farber Cancer Institute, and the Abramson Cancer Center of the University of Pennsylvania are teaming up in a partnership, which will dramatically advance the understanding of the risks associated with multiple cancer genes that are being analyzed through next generation sequencing technology
For more information on PROMPT, please visit https://www.myriadpro.com/for-your-practice/prompt-registry/
Copyright © 2015 Myriad Genetic Laboratories all rights reserved